Biosergen: 1.5 million people die each year from fungal infections. Biosergen, which has FDA orphan drug status, is developing an antifungal drug that is more effective and has a significantly better side-effect profile than existing antifungal drugs developed 40-70 years ago. With USD 16.7 billion spent annually on antifungal drugs, Biosergen’s drug has blockbuster potential (annual sales of more than USD 1 billion) and is expected to be launched in Q2 2026.
Biosergen: Ticker: BIOSGN | Price: SEK 1,318 | Market Cap: 52 MSEK | YTD price performance: 26%
A study that has gone according to plan
Biosergen’s phase 1 study has confirmed that the drug is safe and very important without the same serious side effects as existing drugs. Although the results to date indicated that the study would be successful, the results are a significant price trigger event. The next step is discussions with the FDA about the phase 2 study, where the drug BSG005 will demonstrate its efficacy in diseased patients.
The study included Single Ascending Dose (SAD), where the dose was gradually increased for each new group of subjects, and Multiple Ascending Dose (MAS) where the subjects received the drug intravenously every 7 days with a gradually increasing dose. The study showed that BSG005 had no adverse effects on the kidney or liver. The existing drugs on the market have very harmful effects on the kidney and liver, which is why they are used with caution. Without these side effects, BSG005 has the potential to be the preferred treatment for life-threatening fungal infections, and thus capture a significant share of the approximately USD 17 billion annual market.
Upcoming trigger
- Presentation of data to FDA and discussion of Phase II study Q2 2023
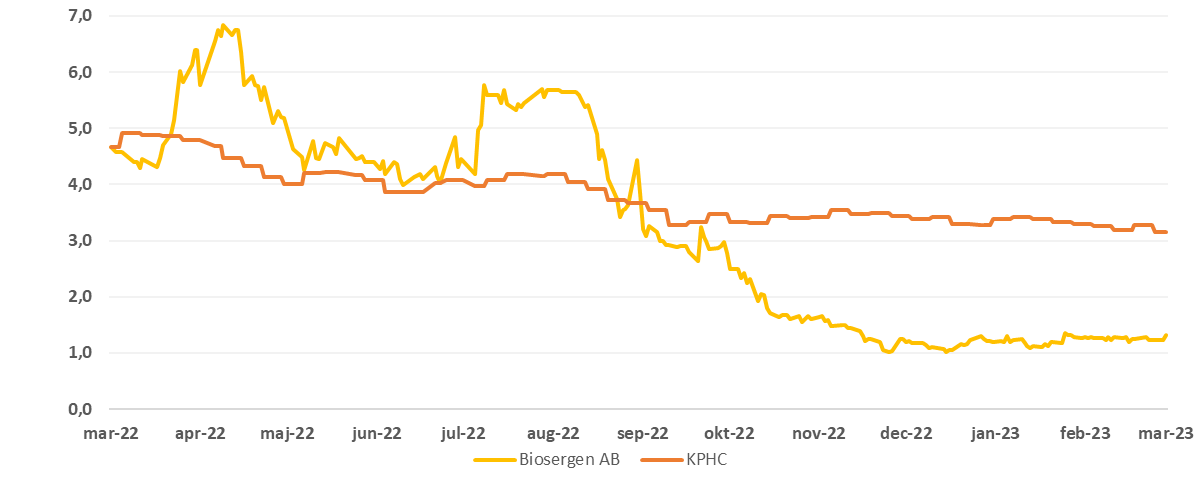
Share for Biosergen the past 1 år vs. Kapital Partner Healthcare Index (KPHC)